@Funkadelic sheeeit, I was an English major. Didn't write this myself, just found it and read it over a couple times. Thanks for the shout out regarding the paint strainer bags, I just so happen to have some on the shelf.
@Ecompost thanks for the insights, sir. Quite thought-provoking as always. Are you positing that the npk shown in the above test results is different than if I mixed chelated minerals to reach the same values? Different in that, because it is derived from Organics, the plant will just take what it needs off of the table (so to speak) whereas with salts it's being force-fed?
Correct me if I'm wrong, but, from what I understand, there is no cut and dry formula for determining the exact available npk from organic additives due to all the variables that make them bio-available. Nothing like hydro buddy or cannastats. I am/was considering finding how many grams are in a 1/2 c of the frass and using that and the above lab analysis to find out precise ppms of the stuff and tailoring the dosage to my nute profile.
I was racking my brain against this idea all day and decided it may be best to simply top dress small amounts of the frass every other week (1tbsp/gal of medium, like with ewc), take notes, and possibly double up the last 3-4 weeks of flower.
I was picturing I'd have to treat frass applications like
MOAB or the like, bumping down the ec of my base fertilizer by half before adding a high pk solution made from soaking the frass.
You are quite right to suggest its virtually impossible to be accurate on PPMs in organics, save to say we have to continue to feed the microbes, and they need to know there is more, or they will start a fight with our plant for the N.
This is why typical regimes are feed all the time. Its less about knowing the PPMS, rather more about knowing your plant has enough when it wants and this will be linked to a very specific exudate and a given plant cycle, and of course enough organic matter, and relevant organisms to process it
even if we take the synth mineral model, then we can only say that we have input xPPMs N, not how much is used, and how much is in NO3- versus NH4+ How much PPM does plant A need, and is this the same as plant B? How much is leaching? How much is binding?
There are charts that help guide us to the optimum PPMS, but in truth we are still guessing or carpet bombing right. Unless we have very high end testing kit, we really are as much in the dark as any organic grower my feel.
In organics, what we can say, organism x say Azotobacter for example, might fix Ykg/Hectare of N, but as you already know this is based on a number of other controlling variables such as kinetics, moisture or other. In the case of Frass, its ability to introduce Chitin based carbohydrates, to indoor gardens so supplementing the availability for microbes to access proteins and enzymes , pivotal in the conversion from organic to ionic N, means you are more likely to maintain a rate of conversion suited to the plant.
This is a pretty good ref on the players etc for say N fixation. I think the point of Organics is to stop worrying if you have the right ingredients at the right time and here in lays one of the many attractions.
On adding Frass, I would only add frass up to week 3 bloom, after which there would little value in its addition imo, this being related to the use of Chtinase via root systems tending to happen from seed up to about day 21 bloom, after which we rarely see Chtin based release and so one might pose the idea its inclusion beyond this point is a waste of energy and money. I use a liquid Chitosan which gives me a wider range of flexibility since i am already aware of the time taken to degrade the matter in this form, I can be more certain of the levels by this key stage marker.

Nitrogen is a critical limiting element for plant growth and production. It is a major component of chlorophyll, the most important pigment needed for photosynthesis, as well as amino acids, the key building blocks of proteins. It is also found in other important biomolecules, such as ATP and nucleic acids. Even though it is one of the most abundant elements (predominately in the form of nitrogen gas (N2) in the Earth’s atmosphere), plants can only utilize reduced forms of this element. Plants acquire these forms of “combined” nitrogen by: 1) the addition of ammonia and/or nitrate fertilizer (from the Haber-Bosch process) or manure to soil, 2) the release of these compounds during organic matter decomposition, 3) the conversion of atmospheric nitrogen into the compounds by natural processes, such as lightning, and 4) biological nitrogen fixation (Vance 2001).
Biological nitrogen fixation (BNF), discovered by Beijerinck in 1901 (Beijerinck 1901), is carried out by a specialized group of prokaryotes. These organisms utilize the enzyme nitrogenase to catalyze the conversion of atmospheric nitrogen (N2) to ammonia (NH3). Plants can readily assimilate NH3 to produce the aforementioned nitrogenous biomolecules. These prokaryotes include aquatic organisms, such as cyanobacteria, free-living soil bacteria, such as
Azotobacter, bacteria that form associative relationships with plants, such as
Azospirillum, and most importantly, bacteria, such as
Rhizobium and
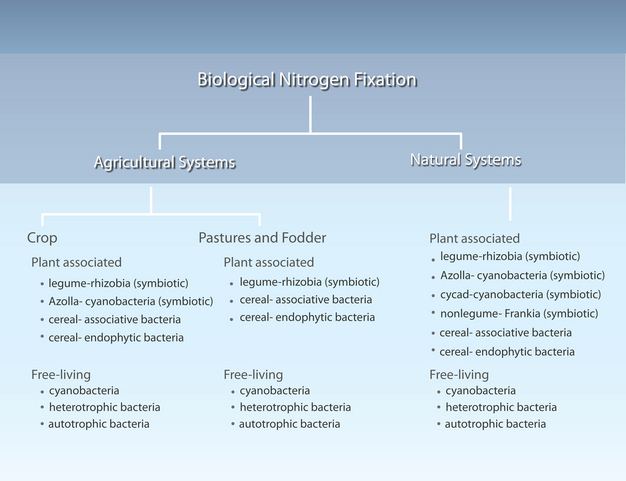
Bradyrhizobium, that form symbioses with legumes and other plants (Postgate 1982). These organisms are summarized in Figure 1.
Figure 1
Nitrogen-fixing organisms found in agricultural and natural systems.
The Process
The reduction of atmospheric nitrogen is a complex process that requires a large input of energy to proceed (Postgate 1982). The nitrogen molecule is composed of two nitrogen atoms joined by a triple covalent bond, thus making the molecule highly inert and nonreactive. Nitrogenase catalyzes the breaking of this bond and the addition of three hydrogen atoms to each nitrogen atom.
Microorganisms that fix nitrogen require 16 moles of adenosine triphosphate (ATP) to reduce each mole of nitrogen (Hubbell & Kidder, 2009). These organisms obtain this energy by oxidizing organic molecules. Non-photosynthetic free-living microorganisms must obtain these molecules from other organisms, while photosynthetic microorganisms, such as cyanobacteria, use sugars produced by photosynthesis. Associative and symbiotic nitrogen-fixing microorganisms obtain these compounds from their host plants’ rhizospheres (National Research Council 1994, Hubbell & Kidder 2009).
Industries use the Haber-Bosch process to reduce nitrogen essentially in the same way. Conventional agriculture has depended upon this process to produce the commercial fertilizer needed to grow most of the world’s hybrid crops. But this approach comes with many consequences, including using fossil fuels for the energy needed to produce this fertilizer, the resulting carbon dioxide emissions and pollution from burning these fuels, and adverse affects on human health (Vitousek 1997).
Overuse of these chemical fertilizers has led to an upset in the nitrogen cycle and consequently to surface water as well as groundwater pollution. Increased loads of nitrogen fertilizer to freshwater, as well as marine ecosystems, has caused eutrophication, the process whereby these systems have a proliferation of microorganisms, especially algae. This “greening” of the water column has caused decreased levels of dissolved oxygen (DO) in bottom waters as planktonic algae die and fuel microbial respiration. These depleted DO levels result in massive mortality of aquatic organisms and create so-called dead zones, areas where little or no aquatic life can be found (Figure 2). Since the 1960’s, dead zones have increased exponentially worldwide, and have now been documented from over 400 systems, affecting more than 245,000 square kilometers of coastal regions (Diaz & Rosenberg 2008, Figure 3). This phenomenon is now deemed the key stressor on marine ecosystems.
more here
http://www.nature.com/scitable/knowledge/library/biological-nitrogen-fixation-23570419