beluga
- 1,532
- 263
Oh, hi, folks!
First and foremost - I should have a general disclaimer attached to all of my posts - they might get verbose!
And this one is not going to be immune to that at all. I will, however, try to pepper in enough eye candy to break up the tedium.
I do these big posts on a desktop, so I also apologize for any formatting that may not be mobile-friendly.
I'm no master guru, but I know how to get it done with some amount of success and reproducibility and I feel like I'm pretty keen to the theory. I hope this can lend something to the community because you all have so much more than I could offer in regard to our usual subject and I've benefited from that greatly. Thank you.
I know the nature of these forums, so don't hesitate to ask questions, post your grows, etc. This shit's free-for-all. But do know that @1diesel1 has already made a general mushroom discussion thread Here. And there's already great stuff and a healthy following there.
This will be my somewhat-personal sister thread to that.
I will head my personal informational posts with:








Cheers!
•••••••••••
•••••••••••
Introduction
So, I've recently been lured back into a passion adjacent to cannabis cultivation... mushroom cultivation!
With (arguably) fewer supplies and negligible energy and space requirements compared to cannabis cultivation, it's a very rewarding and simple hobby to get into.
That said,
I'm not going to show you what the community at large would consider the most basic introductory method - the PF Tek - because I believe it to neglect certain fundamental principles and practices to really be successful and prolific in your fungicultural endeavor.
However, I would be a total asshat to not mention Ziran's Updated PF Tek.
He has outlined and shared an excellent bridging of the gap between the original PF Tek and those aforementioned fundamentals.
If you want to grow mushrooms fast, do that. If you want to learn to cultivate mushrooms, well...
•••••••••••
I hope to make this a guide to get you from point A to point A∞.
From your initial procurement of spores to the innumerable collection of subsequent spores.
Continuity. Perpetuity. The ouroboros. The three hares.
From one compatible set of spores you can have a lifetime of mushrooms.
I also believe in some fundamental principles of education.
Like nomenclature and vocabulary.
Theory before practice.
I want you to have a brief understanding of what it is you're working with before you work with it.
I don't expect people to use technical labels over common ones, but I do believe they are good to know as to assuredly identify what is being described.
•••••••••••
•••••••••••
Glossary of Terms
*You may find some words missing that you think are relevant.
A. I'm not perfec. B. I may have intentionally left them out as to not introduce words that are outside of this method.
A
•••••••••••
•••••••••••
Basic Taxonomy
This is a basidiomycetous Mushroom
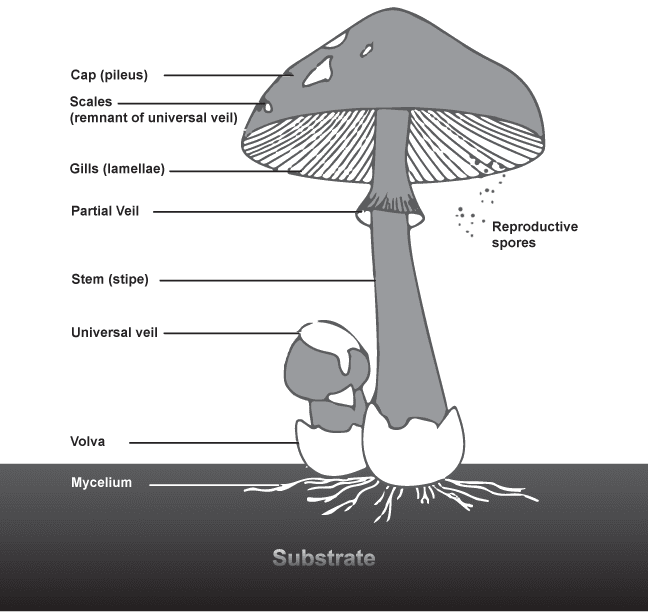
So are P. cubensis!
This is their basic life cycle
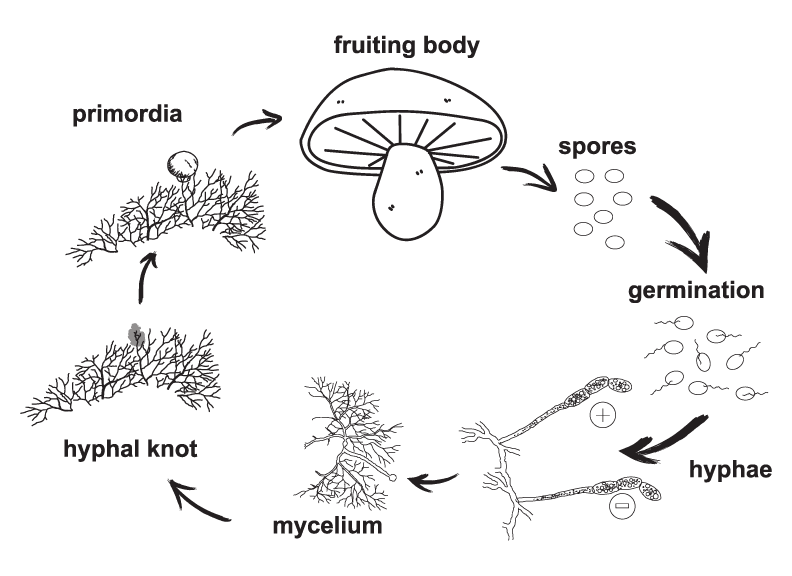
Neat!
•••••••••••
•••••••••••
The Cultivation Process
To cultivate is to promote or improve the growth of a culture by labor and attention.
The basic needs of mushroom growth are air, water, and nutrition.
Inoculation
This is the process of germinating spores or transferring mycelium to a sterile and highly-nutritious spawn substrate.
This spawn substrate is most typically a whole or ground cereal grain such as rye, oat, millet, wheat, or rice.
Colonization
After inoculating, the germinated spores or the transferred mycelium start the vegetative process of growing on the spawn substrate known as colonization.
We sterilize this substrate before inoculating to minimize as much as possible, the opportunity for competing microorganisms, or contamination.
Expansion
Once the spawn substrate is 100% colonized, it is then known simply as spawn. We then expand this spawn to a mildly-nutritious bulk substrate in a process known as spawning.
Properly prepared bulk substrates provide a medium for the mycelium to thrive in which helps maintain adequate hydration (water) and respiration (air). Bulk substrates prepared properly are said to be at field capacity.
Fructification
Or, simply, fruiting is the stage of growth after the mycelium has expanded as far as it can go and is now developing fruiting bodies, or mushrooms. Indoors, this is carried out in a fruiting chamber that simulates natural fruiting conditions - hydration, respiration, and evaporation of the top layer of the colonized bulk substrate, collectively known as surface conditions. Fruiting starts with mycelium joining together in hyphal knots (primordium). Those hyphal knots then go on to develop the first recognizable fruiting bodies, or pins.
Harvest
Once the mushrooms have developed fully, they are preparing themselves for the end of their reproductive cycle - sporulation. Before they open up to spread their spores, their gills are protected by a membranous layer called a veil. Harvest typically happens at or near the time of the veil just starting to tear, before the mushrooms have dropped their spores from their gills.
The other crucial aspect of harvesting is collecting those spores so that we may perpetuate our cultivation. This is done by taking a cap that has not yet dropped its spores to a controlled environment and setting it on a piece of foil or paper to take a spore print.
Preservation
After the mushrooms have been harvested, they must either be used fresh within their short shelf-life or be thoroughly dried and stored in order to preserve them for later use.
The main factors in spoiling of dried fruits are moisture and sunlight, so it is best to store them in an air-tight and opaque container.
•••••••••••
•••••••••••
This Technique
The technique that I'll be using will be, essentially, making clean spawn to expand to a properly hydrated inert bulk substrate in a self-maintaining environment, or monotub.
It's a common one. People have variations on this theme. It is not the end-all-be-all of mushroom cultivation by any means. I did not create it nor do I take credit for any of its processes. I'm just sharing it with you.
Cell Culturing on Agar
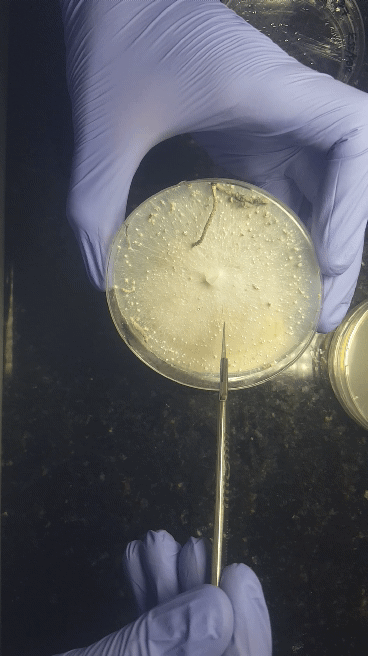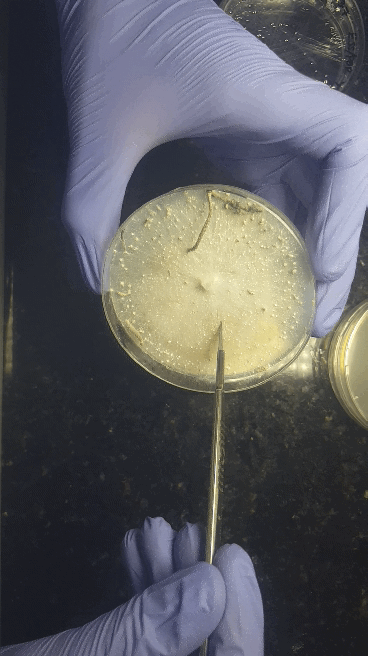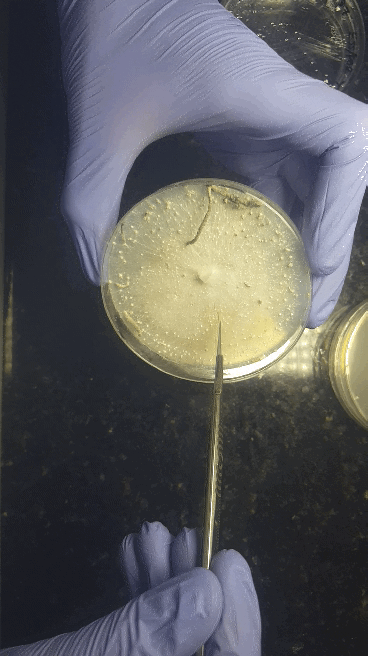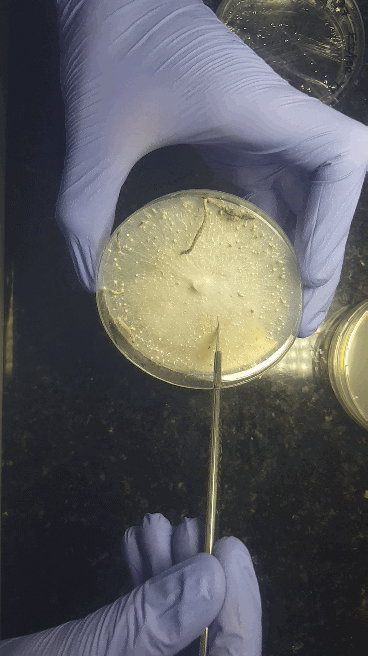
We'll start with bringing spores - either a (multi)spore print or a (multi)spore syringe (MSS) - to a sterilized nutritious agar media on petri dishes or no-pour plates.
Through a series of transfers, we'll learn how to take our desired culture away from any contaminating culture to be left with an axenic culture.
Colonizing Grain Spawn
Once we obtain an axenic culture on agar, we'll next learn to properly hydrate and sterilize grain spawn as to provide optimal conditions for colonization.
Expanding to Bulk Substrates
Once the grain spawn is 100% colonized, we will then learn how to hydrate coir to field capacity. This can be tricky, so I will also suggest the use of vermiculite to help us obtain that field capacity if we happen to oversaturate the coir. After we have our hydrated bulk substrate at field capacity, we will then spawn our 100% colonized spawn to the buk substrate at a ratio ranging from 1:2 - 1:4. For example, 1 quart of spawn to 2 quarts of hydrated bulk substrate.
Maintaining Surface Conditions
Once spawned and mixed with our bulk substrate, we will then simply need to insure that our envrionment is optimal for growing out our mycelium and not encouraging contamination.
This is achieved via adequate fresh air exchange, relative humidity, and temperature. P. cubensis is a very tolerant species and can grow in many ranges. So there is wiggle room, but some extremes can promote contamination.
Harvest, Spore Collection, and Cloning
Once the mushrooms have developed fully, we'll then harvest them (!!!). But we want to make sure we can keep on growing, so we will learn to collect spores with minimal contamination and also learn to clone selected specimens to get the best mushrooms with the most dense canopies.
Preservation and Recipes
After the mushrooms have been harvested, then we eat them!!! But you'll have too many to eat them all within normal parameters of maintaining your sanity. So, we'll go through proper drying, making teas, ice cube cubes, etc. as to store and them keep them from spoiling. Also some added information on MAOI's and things to help make your experience a smoother ride.
•••••••••••
•••••••••••
Supplies
I'll do my best to make a supply list relevant to each section, but I'll also preemptively add everything I can think of here for reference.
Along with these lists, I'll do my best to give you places to get them. I don't know international suppliers at all, so, please bear with me and maybe spark up conversation with your fellow countrymen.
•••••••••••
Supply Key
Follow these symbols to see generally where you could find things.
I would mention online supply/Amazon, but that's kind of a given and, for me at least, generally where I get all my stuff except heavy items like 50lb bags of grain.
Farm & Garden supply -
Laboratory supply -
Medical supply -
Hardware supply - 🛠
Kitchen & Grocery supply -
Big Box/General supply - 🛍
Brewery supply -
Pet supply -
•••••••••••
Agar and Culturing Supplies
Still Air Box - 🛍🛠 • Pressure Cooker - 🛍 • Petri Dishes / Bulk -
• Petri Dishes / Bulk -  • Media Bottle -
• Media Bottle - 
 🛍 • Agar Agar -
🛍 • Agar Agar - 
 🛍 • Malt Extract -
🛍 • Malt Extract - 
 • Torch - 🛠
• Torch - 🛠
 • Scalpel / Blades -
• Scalpel / Blades - 
 • Nitrile Gloves -
• Nitrile Gloves -  🛍🛠
🛍🛠 • 70% Isopropyl -
• 70% Isopropyl -  🛍
🛍
 • Cling Wrap -
• Cling Wrap -  🛍 • Inoculation Loop -
🛍 • Inoculation Loop -  • Synthetic Swabs -
• Synthetic Swabs -  🛍
🛍

•••••••••••
Spawn
Quart Jars - 🛍🛠 • PP5 Lids - 🛍
🛍🛠 • PP5 Lids - 🛍 • Synthetic Filter Discs (SFDs) -
• Synthetic Filter Discs (SFDs) -  • Polyfil - 🛍 • RTV Silicone - 🛠 • Rye -
• Polyfil - 🛍 • RTV Silicone - 🛠 • Rye -

 🛍 • Millet -
🛍 • Millet - 
 🛍 • Wild Bird Seed (WBS) -
🛍 • Wild Bird Seed (WBS) - 
 🛍
🛍
•••••••••••
Bulk Substrate/Habitat
Monotub / Shoebox - 🛍🛠 • Coconut Coir -
 • Spray Mister - 🛍
• Spray Mister - 🛍
 • Vermiculite -
• Vermiculite - 
If you aren't able to find something here - let me know! There can be substitues or different places to procure these things.
For instance, if you can't find SFD's, you can use Polyfil instead. If you have trouble finding Polyfil, well, most pillows with synthetic stuffing are made with it!
There are, however, some things I find absolutely necessary - like a pressure cooker. Most any model will work in some way or another.
•••••••••••
•••••••••••
Alright folks! I think that's about it for my introduction. I sure hope so...
Now, if you want to join along - find some spores! And get everything I have listed under Agar and Culturing Supplies.
I'll give a bit of lagtime to prepare myself and so others who want to follow can prepare themselves. But don't feel like you need to keep up pace... do it at your leisure for your own satisfaction.
Anyone ahead of me, feel free to ask questions, but, again, remember there is The Mushroom Thread. I keep tabs on that and love to advise any way I can there, too.
First and foremost - I should have a general disclaimer attached to all of my posts - they might get verbose!
And this one is not going to be immune to that at all. I will, however, try to pepper in enough eye candy to break up the tedium.
I do these big posts on a desktop, so I also apologize for any formatting that may not be mobile-friendly.
I'm no master guru, but I know how to get it done with some amount of success and reproducibility and I feel like I'm pretty keen to the theory. I hope this can lend something to the community because you all have so much more than I could offer in regard to our usual subject and I've benefited from that greatly. Thank you.
I know the nature of these forums, so don't hesitate to ask questions, post your grows, etc. This shit's free-for-all. But do know that @1diesel1 has already made a general mushroom discussion thread Here. And there's already great stuff and a healthy following there.
This will be my somewhat-personal sister thread to that.
I will head my personal informational posts with:
Cheers!
•••••••••••
•••••••••••
Introduction
So, I've recently been lured back into a passion adjacent to cannabis cultivation... mushroom cultivation!
With (arguably) fewer supplies and negligible energy and space requirements compared to cannabis cultivation, it's a very rewarding and simple hobby to get into.
That said,
I'm not going to show you what the community at large would consider the most basic introductory method - the PF Tek - because I believe it to neglect certain fundamental principles and practices to really be successful and prolific in your fungicultural endeavor.
However, I would be a total asshat to not mention Ziran's Updated PF Tek.
He has outlined and shared an excellent bridging of the gap between the original PF Tek and those aforementioned fundamentals.
If you want to grow mushrooms fast, do that. If you want to learn to cultivate mushrooms, well...
•••••••••••
I hope to make this a guide to get you from point A to point A∞.
From your initial procurement of spores to the innumerable collection of subsequent spores.
Continuity. Perpetuity. The ouroboros. The three hares.
From one compatible set of spores you can have a lifetime of mushrooms.
I also believe in some fundamental principles of education.
Like nomenclature and vocabulary.
Theory before practice.
I want you to have a brief understanding of what it is you're working with before you work with it.
I don't expect people to use technical labels over common ones, but I do believe they are good to know as to assuredly identify what is being described.
•••••••••••
•••••••••••
Glossary of Terms
*You may find some words missing that you think are relevant.
A. I'm not perfec. B. I may have intentionally left them out as to not introduce words that are outside of this method.
A
Agar - an extract from a seaweed used to solidify media
Annulus - the ringlike veil remnant on a mushroom stipe
Axenic - free from other microorganisms; uncontaminated
BBacteria- unicellular microorganisms that may cause contamination in culture work
Basidiomycetes- a group of fungi which produce their spores externally on so-called basidia
Basidium (plural of basidia) - A cell that gives rise to a basidiospore
Biological Efficiency (BE) - A term for yield potential. 100% biological efficiency occurs when 1 lb of fresh mushrooms is harvested from 1 lb of dry substrate, over multiple flushes
Brown Rice Flour (BRF) - ground brown rice
Bruising - a bluing reaction of the mycelium from stress
CCap - see pileus
Casing - a fruiting method where a colonized substrate is covered with a non-nutritious layer
Clone - a population of individuals all derived asexually from the same single parent
Coir - the prepared husk of a coconut fruit, used as a substrate material
Colonization - the stage of growth after inoculation during which the mycelium grows through a spawn substrate
Contamination - undesired foreign material in a substrate, often other infectious microbes
EExpansion - the stage of growth after colonization during which the mycelium grows through a bulk substrate
FField Capacity - content of water in a soil after being saturated and free drainage is negligible
Flame/Torch - a method of sterilization in which a flame or torch is used to bring a metal tool to incandescence
Fresh Air Exchange (FAE) - the amount of air circulation it takes to fruit a given species
Fructification/Fruiting - the state of growth where the mycelium of a colonized substrate produces fruiting bodies, or mushrooms
Fruiting Body - a mushroom; the part of the mushroom that grows above ground
Fruiting Chamber - an enclosed space used to artificially create an environment that simulates fruiting conditions; vivarium
Flush - the phasic even development of multiple fruiting bodies
GGermination - the spreading of hyphae from a spore
Gills - see lamellae
Grain-to-grain transfer (G2G) - The inoculation of grain with already-colonized grain
HHarvest - the gathering of crops ready for consumption or processing
Hypha - a branching filamentous structure of a fungus, multiple hyphae are collectively called mycelium
IIncubation - the period of time after inoculation in which colonization occurs
Inoculation - the process of adding spores or mycelium culture to a substrate
ISO - 70% isopropyl alcohol; a sanitizing agent
Isolate - an uncombined fungal strain
LLamellae - the tiny segments on the underside of the pileus where spores are produced
Light Malt Extract (LME) - a powder of malted barley typically used in brewing beer
Liquid culture (LC) - A culture of mycelium suspended in a nutritious liquid, for use as an inoculant
Liquid Inoculant (LI) - a suspension of mycelium in water. This is different from a LC in that the mycelium did not grow in the medium
MMalt Extract Agar (MEA) - a nutritious culturing medium that combines water, agar, and malt extract
Millet - a small cereal grass grain used as a spawn substrate
Monotub - a clear container used as fruiting chamber
Multispore (MS) - refers to an inoculation where multiple germinations and matings occur due to the use of various spores, as in a spore solution (e.g. spore syringe) and as opposed to an isolate
Multispore syringe (MSS) - see spore syringe
Mycelium- the portion of the mushroom that grows underground
Mycorrhiza - a symbiotic association between a plant root and fungal hyphae
PPartial Veil - a membranous covering reaching from the stipe to the pileus of a mushroom to protect spores developing in the lamellae
Petri dish - a round glass or plastic dish with a cover to observe the growth of microscopic organisms
Pileus - the horizontal portion of a mushroom; cap
Pin - a term used to describe a very young mushroom
Polyfill- a polyester fiber used for gas exchange and filtering microorganisms
Preservation - the process of preparing food or other perishables to resist spoilage or decomposition
Pressure Cooker (PC) - a pot with a tight-fitting lid that uses steam and high pressure to cook and preserve foods, used for sterilization
Primordium - the initial fruiting body, sometimes referred to as a ‘hyphal knot’
RRelative Humidity (RH) - the amount of water vapor in the air
Rhizomorphous (rhizo) - used to describe a rootlike characteristic of mycelium growth
RTV Silicone - Room Temperature Vulcanizing silicone. Used as a high-temperature gasket and adhesive
Rye - a cereal grass grain used as a spawn substrate
SShoebox - a small monotub
Spawn - a culture of mycelium on a substrate
Spore - the reproductive unit of a fungus
Spore Print - a collection ofspores taken from a mushroom cap, often collected on sterile card stock, aluminum foil, or some other flat surface
Spore Syringe - a collection of spores suspended in water in a syringe
Sporulation - when a mature mushroom releases its reproductive spores
Stem - see: stipe
Sterile Technique - a method of careful manipulations that prevents foreign cells from entering cultures or media
Sterilization - the procedure of destroying all living organisms in or on a given
Still Air Box (SAB) - a mostly-sealed transparent container designed to allow one to manipulate objects in an environment without air currents
Stipe - the stem or stalk-like feature supporting the pileus of a mushroom
Strain - the result of compatible spores mating; there are innumerable potential strains in a single mushroom's sporulation
Substrate (sub) - anything used to grow mushrooms on
Surface Conditions - the moisture content and rate of evaporation on the top of a substrate or casing
Synthetic Filter Disc (SFD) - a woven cellulose or PTFE material used for gas exchange and filtering microorganisms
TTek - shorthand for technique
Tissue culture (TC) - a sample of tissue used to culture a genetic duplicate; clone
Tomentose - used to describe a fluffy characteristic of mycelium growth
Transfer (xfer) - used to describe taking a small piece of a culture from one culturing media to another
UUniversal Veil - temporary membranous tissue that envelopes immature fruiting bodies, often leaving remnants on the pileus
VVariety - a distinct group of organisms with a set of unique and limited genetic traits; race (obsolete)
Veil - see partial or universal veil
Vermiculite - a highly absorbent substrate material made from puffed mica
WWBS - Wild bird seed. Millet-based grains used as a spawn substrate
•••••••••••
•••••••••••
Basic Taxonomy
This is a basidiomycetous Mushroom

So are P. cubensis!
This is their basic life cycle

Neat!
•••••••••••
•••••••••••
The Cultivation Process
To cultivate is to promote or improve the growth of a culture by labor and attention.
The basic needs of mushroom growth are air, water, and nutrition.
Inoculation
This is the process of germinating spores or transferring mycelium to a sterile and highly-nutritious spawn substrate.
This spawn substrate is most typically a whole or ground cereal grain such as rye, oat, millet, wheat, or rice.
Colonization
After inoculating, the germinated spores or the transferred mycelium start the vegetative process of growing on the spawn substrate known as colonization.
We sterilize this substrate before inoculating to minimize as much as possible, the opportunity for competing microorganisms, or contamination.
Expansion
Once the spawn substrate is 100% colonized, it is then known simply as spawn. We then expand this spawn to a mildly-nutritious bulk substrate in a process known as spawning.
Properly prepared bulk substrates provide a medium for the mycelium to thrive in which helps maintain adequate hydration (water) and respiration (air). Bulk substrates prepared properly are said to be at field capacity.
Fructification
Or, simply, fruiting is the stage of growth after the mycelium has expanded as far as it can go and is now developing fruiting bodies, or mushrooms. Indoors, this is carried out in a fruiting chamber that simulates natural fruiting conditions - hydration, respiration, and evaporation of the top layer of the colonized bulk substrate, collectively known as surface conditions. Fruiting starts with mycelium joining together in hyphal knots (primordium). Those hyphal knots then go on to develop the first recognizable fruiting bodies, or pins.
Harvest
Once the mushrooms have developed fully, they are preparing themselves for the end of their reproductive cycle - sporulation. Before they open up to spread their spores, their gills are protected by a membranous layer called a veil. Harvest typically happens at or near the time of the veil just starting to tear, before the mushrooms have dropped their spores from their gills.
The other crucial aspect of harvesting is collecting those spores so that we may perpetuate our cultivation. This is done by taking a cap that has not yet dropped its spores to a controlled environment and setting it on a piece of foil or paper to take a spore print.
Preservation
After the mushrooms have been harvested, they must either be used fresh within their short shelf-life or be thoroughly dried and stored in order to preserve them for later use.
The main factors in spoiling of dried fruits are moisture and sunlight, so it is best to store them in an air-tight and opaque container.
•••••••••••
•••••••••••
This Technique
The technique that I'll be using will be, essentially, making clean spawn to expand to a properly hydrated inert bulk substrate in a self-maintaining environment, or monotub.
It's a common one. People have variations on this theme. It is not the end-all-be-all of mushroom cultivation by any means. I did not create it nor do I take credit for any of its processes. I'm just sharing it with you.
Cell Culturing on Agar
We'll start with bringing spores - either a (multi)spore print or a (multi)spore syringe (MSS) - to a sterilized nutritious agar media on petri dishes or no-pour plates.
Through a series of transfers, we'll learn how to take our desired culture away from any contaminating culture to be left with an axenic culture.
Colonizing Grain Spawn
Once we obtain an axenic culture on agar, we'll next learn to properly hydrate and sterilize grain spawn as to provide optimal conditions for colonization.
Expanding to Bulk Substrates
Once the grain spawn is 100% colonized, we will then learn how to hydrate coir to field capacity. This can be tricky, so I will also suggest the use of vermiculite to help us obtain that field capacity if we happen to oversaturate the coir. After we have our hydrated bulk substrate at field capacity, we will then spawn our 100% colonized spawn to the buk substrate at a ratio ranging from 1:2 - 1:4. For example, 1 quart of spawn to 2 quarts of hydrated bulk substrate.
Maintaining Surface Conditions
Once spawned and mixed with our bulk substrate, we will then simply need to insure that our envrionment is optimal for growing out our mycelium and not encouraging contamination.
This is achieved via adequate fresh air exchange, relative humidity, and temperature. P. cubensis is a very tolerant species and can grow in many ranges. So there is wiggle room, but some extremes can promote contamination.
Harvest, Spore Collection, and Cloning
Once the mushrooms have developed fully, we'll then harvest them (!!!). But we want to make sure we can keep on growing, so we will learn to collect spores with minimal contamination and also learn to clone selected specimens to get the best mushrooms with the most dense canopies.
Preservation and Recipes
After the mushrooms have been harvested, then we eat them!!! But you'll have too many to eat them all within normal parameters of maintaining your sanity. So, we'll go through proper drying, making teas, ice cube cubes, etc. as to store and them keep them from spoiling. Also some added information on MAOI's and things to help make your experience a smoother ride.
•••••••••••
•••••••••••
Supplies
I'll do my best to make a supply list relevant to each section, but I'll also preemptively add everything I can think of here for reference.
Along with these lists, I'll do my best to give you places to get them. I don't know international suppliers at all, so, please bear with me and maybe spark up conversation with your fellow countrymen.
•••••••••••
Supply Key
Follow these symbols to see generally where you could find things.
I would mention online supply/Amazon, but that's kind of a given and, for me at least, generally where I get all my stuff except heavy items like 50lb bags of grain.
Farm & Garden supply -
Laboratory supply -
Medical supply -
Hardware supply - 🛠
Kitchen & Grocery supply -
Big Box/General supply - 🛍
Brewery supply -
Pet supply -
•••••••••••
Agar and Culturing Supplies
Still Air Box - 🛍🛠 • Pressure Cooker - 🛍
•••••••••••
Spawn
Quart Jars -
•••••••••••
Bulk Substrate/Habitat
Monotub / Shoebox - 🛍🛠 • Coconut Coir -
If you aren't able to find something here - let me know! There can be substitues or different places to procure these things.
For instance, if you can't find SFD's, you can use Polyfil instead. If you have trouble finding Polyfil, well, most pillows with synthetic stuffing are made with it!
There are, however, some things I find absolutely necessary - like a pressure cooker. Most any model will work in some way or another.
•••••••••••
•••••••••••
Alright folks! I think that's about it for my introduction. I sure hope so...
Now, if you want to join along - find some spores! And get everything I have listed under Agar and Culturing Supplies.
I'll give a bit of lagtime to prepare myself and so others who want to follow can prepare themselves. But don't feel like you need to keep up pace... do it at your leisure for your own satisfaction.
Anyone ahead of me, feel free to ask questions, but, again, remember there is The Mushroom Thread. I keep tabs on that and love to advise any way I can there, too.
Last edited: