I don't know what to say I am speechless, oil really! Is that Italian or extra light or canola , boggles the mind. Using oil in your hydroponic system is only for vegetables!. ;) ..I have never heard such a preposterous idea. You sure he's your friend? :eek:
There is just so much wrong with that idea, stuff dies when coated with oil, umm like roots, oil harbors and attracts things, you would never get the system clean.
It's a good thing your getting a second opinion. :)
I DO NOT THINK THAT IS A GOOD IDEA, what greeneye04 ^^^^ said makes more sense. You got to send that question to Daniel at UC HQ o_O
Peace
I would def,leave the oil for cleaning scissors and hands works great for that
Actually, while this might seem quite silly--as it is not what you are used to doing with oil. Oil works quite well at destroying most bacteria.
Some gram positive bacteria will survive, but these tend to be the types of bacteria that attack us--not our plants.
What happens here is very simple.
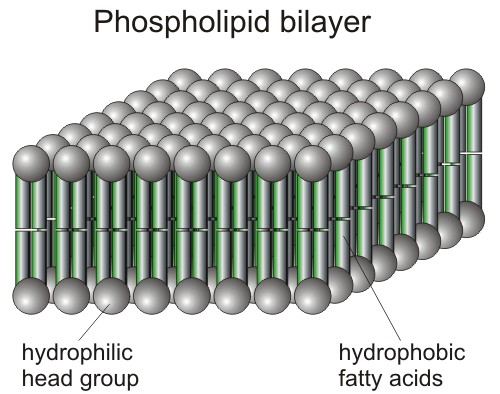
The bacteria possesses an outer membrane (they are single celled organisms). This membrane is known as a phospholipid bilayer.
It is made up of what it sounds like (and I'll oversimplify it here for the sake of this discussion) A lipid tail and a phosphate "head".
The phosphate is polar and interacts well with water (in the extracellular medium and in the intracellular cytosol). The lipid "tails" prefer to hang out together--as a result of something known as hydrophobic interactions.
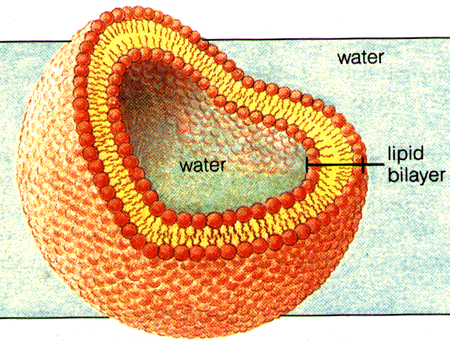
In fact, if you throw a bunch of phospholipids into water, lipid bilayers (spherical ones, known as miscelles) will form spontaneously.
When we add vegetable oil (which consists mostly of polyunsaturated triacylglycerols--or triglycerides)--the fat molecules (and other molecules present, such as other phospholipids) can start to insert into the lipid bilayer (fatty acid end first).
This destabilizes the membrane and can cause one of a few deaths for the bacterium:
1. Bi-layer collapses and the organism is destroyed.
2. Membrane becomes too large--making it impossible for the bacterium to maintain necessary charge/ion gradients for metabolism.
3. Membrane becomes too "soft", the bacterium will die when it bumps into something--and its ability to thrive in the current temperature (and higher temperatures) will be significantly reduced.
4. Membrane becomes too rigid and the opposite will happen--i.e. lower temperatures will kill the bacterium.
This is FAR FAR FAR from a 100% solution, but it should work in a great deal of cases.
So says science.
P.S. this should also be effective on a variety of:
1. Endospores.
2. Fungal spores
3. Insect eggs/larvae