Frankster
Never trust a doctor who's plants have died.
Supporter
- 5,188
- 313
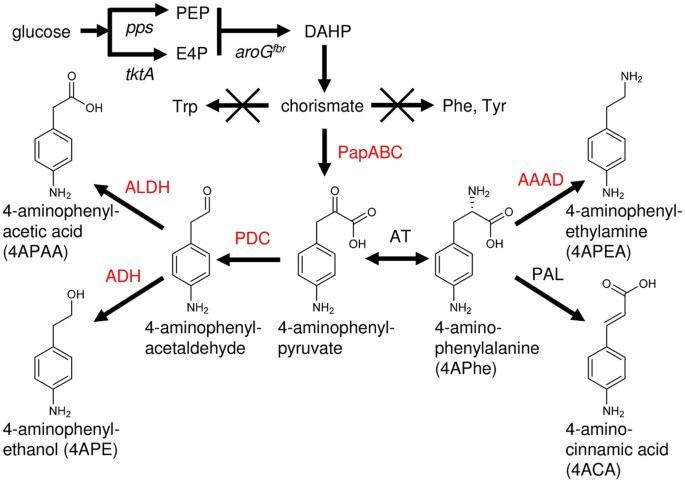
Can aromatic amines be utilized to fuel and direct plant development?
In organic chemistry, amines are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia, wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group.
In chemistry, aromaticity is a property of cyclic (ring-shaped), planar (flat) structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to other geometric or connective arrangements with the same set of atoms. Aromatic rings are very stable and do not break apart easily. Organic compounds that are not aromatic are classified as aliphatic compounds—they might be cyclic, but only aromatic rings have enhanced stability.
Since the most common aromatic compounds are derivatives of benzene ,the word aromatic occasionally refers informally to benzene derivatives, and so it was first defined. Nevertheless, many non-benzene aromatic compounds exist. In living organisms, for example, the most common aromatic rings are the double-ringed bases in RNA and DNA. An aromatic functional group or other substituent is called an aryl group.
An aromatic amine is an organic compound consisting of an aromatic ring attached to an amine. It is a broad class of compounds that encompasses anilines, but also many more complex aromatic rings and many amine substituents beyond NH2. Such compounds occur widely
aka: offering up to the plant various building blocks of life, during key times in development.
In organic chemistry, amines are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia, wherein one or more hydrogen atoms have been replaced by a substituent such as an alkyl or aryl group.
In chemistry, aromaticity is a property of cyclic (ring-shaped), planar (flat) structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to other geometric or connective arrangements with the same set of atoms. Aromatic rings are very stable and do not break apart easily. Organic compounds that are not aromatic are classified as aliphatic compounds—they might be cyclic, but only aromatic rings have enhanced stability.
Since the most common aromatic compounds are derivatives of benzene ,the word aromatic occasionally refers informally to benzene derivatives, and so it was first defined. Nevertheless, many non-benzene aromatic compounds exist. In living organisms, for example, the most common aromatic rings are the double-ringed bases in RNA and DNA. An aromatic functional group or other substituent is called an aryl group.
An aromatic amine is an organic compound consisting of an aromatic ring attached to an amine. It is a broad class of compounds that encompasses anilines, but also many more complex aromatic rings and many amine substituents beyond NH2. Such compounds occur widely
aka: offering up to the plant various building blocks of life, during key times in development.
Representative aromatic amines | ||
| Aromatic ring | Name of parent amines | Example |
| benzene | aniline | substituted anilines |
| benzene | phenylenediamines | the antioxidant p-phenylenediamine |
| toluene | toluidines | the pharmaceutical prilocain |
| toluene | diaminotoluenes | the hair dye ingredient 2,5-diaminotoluene |
| naphthalene | naphthylamines | the dyes Congo red and Prodan |
| pyridine | aminopyridines | the drug tenoxicam |
| pyrimidine | aminopyrimidines | the nucleobase cytosine |
| quinoline | aminoquinolines | the drug primaquine |
| purine | aminopurines | the nucleobase guanine |
| acridine |