Definitions:
Strain: A genetically distinct member of the species, which can be a monokaryon, or a dikaryon. Every spore in a print yields a genetically distinct monokaryon, so every spore could be said to contain a strain.
My usage of the word here is mostly referring to dikaryotic strains capable of fruiting.
Nucleus(ei): The best part of a cell, which contains all of the DNA. It houses all the genetics of an organism, and ultimately controls every single process or trait. Tasty, but you need a lot of them for a good meal.
Plasmogamy: The open exchange of cytoplasm and cellular contents between two cells, preceded by anastomosis. For fungi, it represents fusion and exchange of nuclei.
Anastomosis: The fusion of two hyphae, technically the term refers to the fusion of two tubes.
Mono-karyon: Single-Seed, a fungal colony where each cell only contains a single set of the genome, haploid.
Di-karyon: Two-seed, a fungal colony where each cell contains two sets of the genome, somewhat similar to diploidy.
Spore: The propagule formed by a fungus, in this case a sexual spore produced by a mushroom. It is haploid, containing a single set of genetics, and germinates to form a monokaryon.
Mushroom: Seriously?
Colony: A single individual, the collective reference to a single cohesive strain in the form of mycelium. There can be multiple colonies of the same strain, but not multiple strains of the same colony.
Meiosis: The fusion of two unique nuclei, followed by segregation, recombination, and division into new unique nuclei that contain genetics from both parents, but in new combinations. Mutation can produce novel genes possessed by neither parent. See; SEX (but not mating, which is separate in fungi).
FIRST PAGE
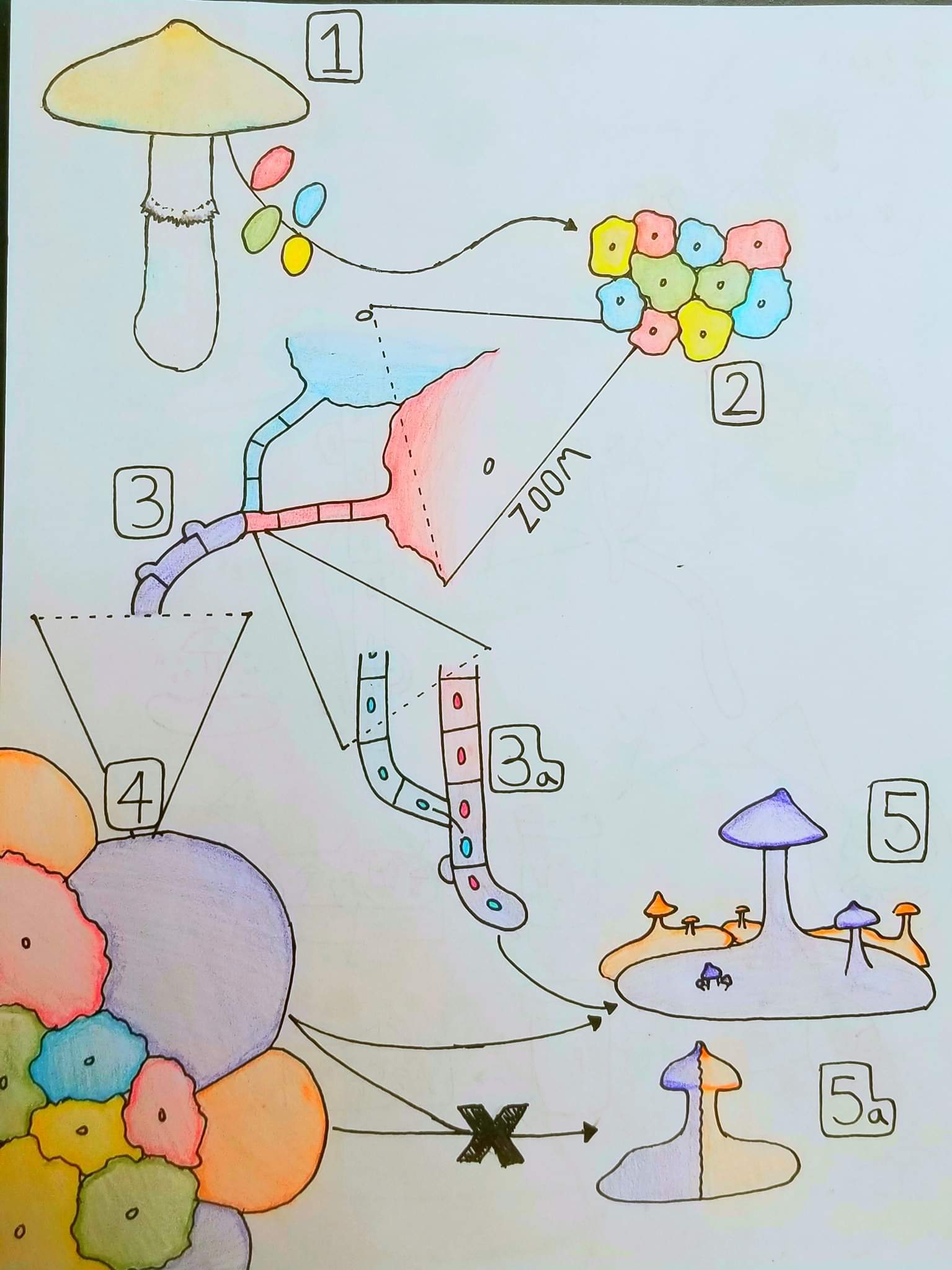
1:
A mushroom releases spores, each of varying genetics and mating type. Each spore is represented by a different color, R, B, G, or Y. A single spore is haploid, carrying only a single set of the genome.
2:
The spores germinate as a group, forming multiple colonies that also only possess a single set of the genome, making them “monokaryotic”, and making the colonies “monokaryons”.
3:
If compatible, the hyphae of two monokaryons will fuse into a new colony, possessing both of their sets. This colony is “dikaryotic”, and is called a “dikaryon”. It is illustrated as purple, being a fusion of the red and blue monokaryons.
3a:
An illustration of the transfer of genetic material:
Each cell in the monokaryon contains just a single nucleus of identical genetics. Upon fusion (anastomosis), one of the colonies (red in this case) will donate nuclei through a portal formed by their fusion (plasmogamy). The new cells containing both red and blue nuclei are dikaryotic, and behave much differently from monokaryons. The cells are wider, grow faster, and form clamp connections which coordinate the pairing of the nuclei into new cells. Now that two sets of genetic material are present, sex (meiosis) is possible.
4:
The dikaryons are illustrated as purple and orange, for convenience, though in reality it would be a rainbow of colors, one for each dikaryon. Not all purple or orange colonies are the same genetically, each one represents a unique dikaryon.
Dikaryons grow way faster than monokaryons, and as soon as they are formed, start to outgrow their parent colonies. The dikaryons are incompatible and stay separate from one another, unlike monokaryons they will not fuse or exchange genetics. Now that their goal is fruiting, it is a territory dispute, rather than a fuckfest.
They radiate away from the original group of monokaryons.
5:
Each of these dikaryons will grow and dominate territory until enough resources have been accumulated that it decides to fruit (ignoring the many environmental factors involved). The mycelium of the dikaryotic colony goes through a series of changes, and through the process of primordial initiation. The mycelium making up the mushroom is identical in every way to the mycelium it came from, it is all the same purple dikaryotic cells.
Even if there are multiple dikaryons overlaid and directly involved with one another, their genetics stay separate, and fruitbody formation is performed only by a single colony without involvement from any other. At this point in time we are far from the point where genetics are flexible.
5a:
An example of what does not happen. Two dikaryons, two strains, will not work together to produce a fruitbody, no matter how many of them are present and overlapping. It is hard enough for a single colony to coordinate with itself to produce such a complex structure, let alone for two unrelated colonies (who want eachother dead) to work together and make it happen. This is not even really up for debate, it is just a basic fact of fungal biology. Some oddballs like Armillaria have segregate tissues made up of haploid, dikaryotic, and diploid mycelia, but the vast majority of agaricoid species (including Psilocybe) have homogenous tissue, each cell being identical (except for the basidia, which are briefly diploid, and the spores, which are haploid).
NEXT PAGE
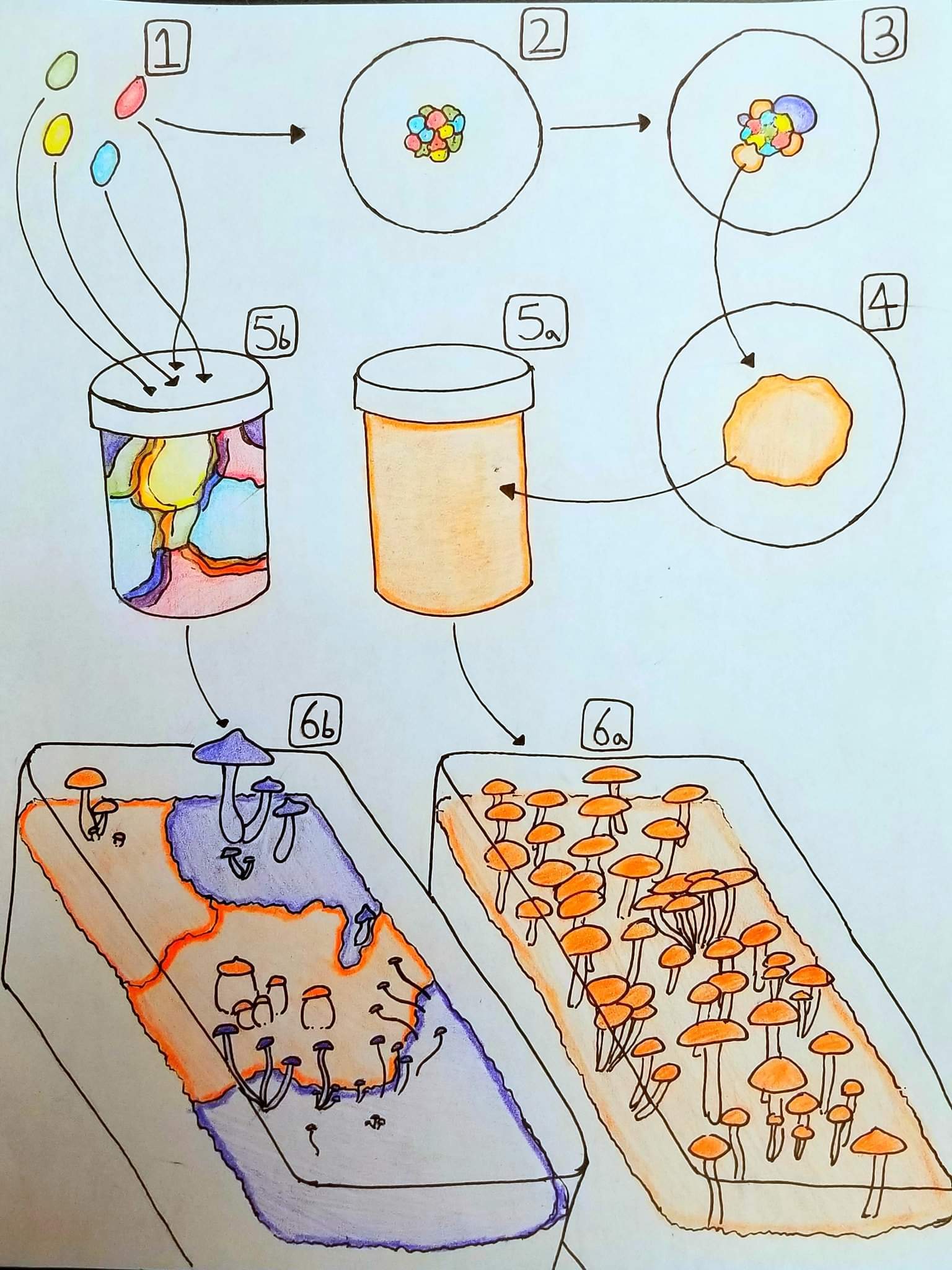
1:
In cultivation, spores are used a few ways, I’ll show a couple.
2:
The spores are placed on agar, usually through a spore solution or scrape. These spores germinate to form monokaryons. Visually, this is the first growth you’ll see, forming a small patchy group extending the area you inoculated.
3:
Eventually, the compatible monokaryotic colonies will start fusing with eachother, and forming dikaryons. On agar, these manifest as a sudden increase in growth rate starting at the edge of the area inoculated. In my experiments, the monokaryons don’t actually grow very far beyond the area you inoculate, the first growth to start dramatically extending past it is all dikaryotic.
It does depend on the amount of spores you used, but in almost all scenarios a very large amount of dikaryons are going to form somewhat simultaneously, all trying to get as much colonized as they can. Rapidly, the ones most capable of outcompeting (which may or may not be correlated with growth rate) sort out and the number of active dikaryons drastically reduces. You won’t really see the massive amount of dikaryons formed, because most of them never get very big. You only see the successful ones.
As they get bigger, growth rate or appearance will make them noticeably different from one another, they’ll form what people call sectors. There is some level of overlap, but the very front margin of a sector should be almost entirely a single colony.
4:
With proper isolation methods, you can separate one of the dikaryotic sectors and “isolate” it away from all of the others (I’m not sure why people need to make 4 damn transfers, you can do it in a single step). This produces a single colony, the same all around. These are not always symmetrical, not always even, and the growth rate and morphology can change over time.
5a:
You then inoculate spawn or inoculant with this isolate, allowing inoculation with a single strain.
5b:
Spores added directly to spawn or inoculant, where steps 2-3 take place uncontrolled within the substrate, skipping step 4 altogether. This means that you can have many many dikaryons colonizing the substrate at once, competing for space and resources.
6a:
Substrate inoculated with a single colony will colonize quickly and contain the same genetics throughout, it will be totally clonal. The fruits produced by this colony will all be the same genetically, cloning from any one of them will yield the same exact isolate, identical to the one in step 4.
Because the substrate is colonized by a single colony, all of the resources have been available and utilized towards its growth, and fruiting takes place more or less evenly across the entire surface, cooperatively.
6b:
Substrate inoculated with multiple dikaryons will often divide into a patchwork, as each one grows and walls the others off. Because they do not cooperate, the substrate gets divided up into territories, and each one only gets a small portion of the resources, rather than all of them.
Each colony will fruit on its own timeline, with its own traits, separate from the others. You can get multiple phenotypes or “varieties” in a single tub/fruiting. In this scenario, cloning different mushrooms may well get you genetically distinct isolates, unless you accidentally clone two from the same colony.
The reduced amount of available resources means that each colony is only producing a small portion of what it could given the whole tub. Fruiting tends to be sparse and inconsistent, with smaller overall fruits. In terms of outright yield, this method tends to be lower, and less consistent.
Even though there are multiple colonies overlapping, multiple colonies fruiting, you cannot get multiple strains from the same mushroom. Each one still only contains mycelium from the colony that made it, and that colony is a single dikaryon with a set amount of genetics. There is only one individual there.
CONCLUSION
The main takeaway is that while yes, there are technically two strains comprising a dikaryon, two strains inside a fruitbody, they ceased to exist as individuals in step 3 on the first page. From that moment on there is only a single cell type in the mycelium, and the fruitbody is only made of that mycelium. Thus, cloning a mushroom from any source, any place or origin, will yield only a single dikaryotic strain per mushroom.
I think people see asymmetrical results on their first tissue culture, and think it represents multiple genetics.
There are a few reasons for that.
The trauma of transfer means that not all sides of the tissue will jump off equally, one side tends to do better than the others, making a large “sector” appear surrounded by sparse young growth.
Not all colonies are symmetrical, even under ideal conditions, some isolates just grow with uneven margins or dead zones. Light can play a role in this, as is seen in Schizophyllum commune and a few other species.
Occasionally, and especially with newer people, spore contamination can occur, producing actually different strains on the tissue clone plate. These should be small and short lived, generally not an issue, but with proper technique (or collecting before sporulation), it should not be occurring.
Genetic damage can occur from the trauma of cloning, potentially producing new colony types or faulty looking sectors.
Sometimes there isn’t an explanation, and what appear to be sectors are present. This does not justify the prospect of there being multiple strains in a fruitbody. There is just so much evidence, such a large amount of precedent showing that this isn’t the case. Whatever may be going on, it is not the presence of multiple strains. Every single hyphae showing up on that plate came from the same genetic source, the same individual