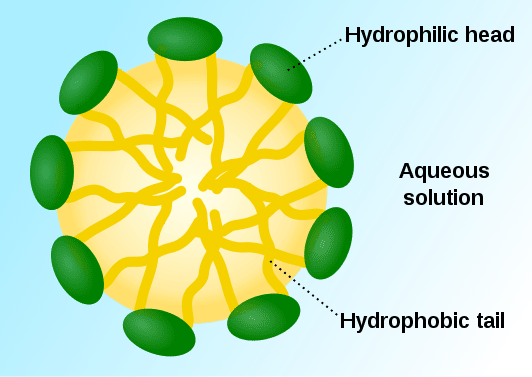
Bubbles form in structures known as micelles which are arranged thusly:
This orientation can be flipped if they are in a nonpolar solvent like hexanes.
As Seamaiden suggests this has to do with polarity.
Hydrophilic essentially means "polar", whereas hydrophobic is nonpolar.
This arrangement of molecules is, while the least thermodynamically "expensive" formation for this type of molecule in a polar solvent, actually a fairly ordered structure. Because of entropy these molecules will not tend to arrange themselves this way if there is no polar solvent present.
What happens when you skim the foam off is that basically, air is a nonpolar "solvent" for the most part--but its not extremely polar either. It's in a bit of a middle ground. So there is no reason for the bubbles to keep their shape.
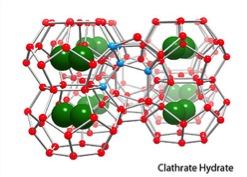
Essentially the only reason that the bubbles form in the first place is that it would be more difficult for the water to create complex structures around the nonpolar ends of these molecules. These structures are typically called clathrates, and they are even MORE ordered than micelles. Because entropy basically says that the least ordered thermodynamic configuration will be favored you end up with micelles rather than clathrates. In a strange way (and this isn't always clear without a lot of exposure to theory) it's actually the existence of the possibility for clathrates that drives the formation of micelles. I can't really demystify the reasoning behind that without a bunch of math and a few hours of lessons lol.
When you take the foam out of solution the compound/mixture has no need to arrange itself into either of these configurations and so it just falls apart (probably into a liquid).
As an experiment you can take this liquid, reintroduce it into a cup of water and whip it up really well--you should end up with bubbles.
Another possibility is that the inside of the bubbles is actually made up of, mostly nonpolar, air and that you're getting some thermodynamic interactions from that and the formation of bubbles has more to do wit viscosity/polarity of the liquid itself than it does with amphpathic molecules in it.
Clathrates look something like this: