B
BUDOGHI
- 85
- 8
I'm searching for CBD rich strains in seed form can any one direct me to mail order availability? I have a friend who benefits from cannabis but does not like the high aspects of the medicine.

can you please tell me more about these! havent seen anyone else here growing them but the clubs especially harborside just eat up the high CBDIm growing Sour Tsunami seeds that I got from Lawrence Ringo and Southern Humboldt seed co.

http://sohumseeds.com/INDEX.html
And Harlequin clones I got from Harborside Heath center in Oakland Ca.
Both are High in CBD
Check out http://www.projectcbd.com/
March 01 Sour Tsunami Stabilized
For the first time, a California grower has "stabilized" a Cannabis strain high in Cannabidiol. Lawrence Ringo of the Southern Humboldt Seed Collective is now offering seeds of "Sour Tsunami" that have a one-in-four chance of containing 10-11% CBD (and 6-7% THC)!
Ringo, 54, has been dealing with severe back pain since early adolescence. "I have no disk between my lower third and fourth," he told us. "It’s bone on bone. When I was 13 some kids thought it was funny to let me take the weight of this thing we were lifting up. I‘ve been dealing with the pain ever since. I’ve been to every doctor and chiropractor. No one can really help. There’s no way that I’m going to do any drugs, ever. I despise them. And I'm not going to do any back surgery. I'm going to rely on the strains I’ve developed over the years. When my back really hurts I do the the high-CBD kief. Two pipeloads and I can go out and do anything —ride a motorcycle, work in the garden."
click for larger image
Ringo started growing at 15. On his way to school one day he found a sapphire gold ring and a snub-nosed .38 and a film can of cocaine. He threw away the coke and returned the ring and the pistol to the local head of the Hell's Angels. "Come on in little brother," said the grateful biker. When asked what he wanted way of thanks, Lawrence pointed to a big Sativa plant he saw growing in the backyard and said, "I want to learn how to grow that!"
At age 19 he went to college Solano Community College and learned organic horticulture. He has been crossing plants of interest for many years, with the goal of creating a strain that would lessen his back pain without giving him "a clogged head." He says, "There are certain highs that you can operate on and some that you can't. You smoke three hits and you’re passed out. I don’t like that."
"I saved seeds whenever a plant gave me a creative feeling," he explains. "I bought 'New York City Diesel' seeds from Marc Emery. I liked it —great taste and no couch lock. I took the males from that and crossed them onto Sour Diesel female clones. I crossed the NYC Diesel and the Sour Diesel for about four years kept crossing it back to the Sour Diesel clone. Each year I’d save the male and do the dusting and make my seeds. One year a friend popped in with something that had a really nice creamy taste. I thought it was an Indica but it turned out to be 60% Sativa called Ferrari. I’ll never forget that creamy taste. I smoked it and my brain just took off. I’m a musician so I just grabbed a guitar and was going nuts for hours."
"It took me about a year to get that Ferrari clone. It was protected by the rednecks — the good old boys who used to be loggers and now they’re pot growers. They have this little clique going, the good old boys network. Very secretive and protective. They weren’t going to give it to us but we got it. I crossed my Sour Diesel —Double Diesel, I called it—to the Ferrari clone five years ago. Two years ago I re-crossed it back onto the Sour Diesel. Before I did that I had this thing called Tsunami. It was the Double Diesel crossed four times to the Sour Diesel then crossed to the Ferrari and then crossed back to the Sour Diesel. That made Sour Tsunami. It's unbelievable. The creamy taste was there. I had no idea what the CBD count was until Samantha Miller [of Pure Analytics] analyzed my stuff and said it was 11.3% CBD. 'And by the way,' she said, 'You have eight other strains with great CBD potential.'"We asked Miller to elaborate. She explained, "So far we have tested 14 of Ringo's strains and 9 of them have shown at least low, if not moderate-to-high amounts of CBD. This is an indicator of the genotype of the plant, that it possesses the required form of a particular gene needed for CBD synthesis. Seeing this indicator tells us that the sibling seeds of the plants that produced the high CBD also have the potential. What we do not know is: do they have potential for moderate-to-high amounts of CBD? Thus these sibling seeds are good targets for potential high-CBD genetics. It also tells us that if we identify two plants with CBD potential, we may inbreed them with a high likelihood of a predictable 1:2:1 result for high CBD, moderate CBD, and no CBD. Thus the progeny from these plants and in-breeding represent targets with a high statistical probability of being high-CBD- producing plants. I say plants and not strains, intentionally."
click for larger image


nice dude! looking great!and Here is a little Sour Tsunami clone (#1A) I was testing for sex...
and I just let it keep flowering..
Its almost ready.. and then I may have it tested for CBD content
nice dude! looking great!
cant wait to see a final smoke report!!
imo i think its a must to get these tested... iirc the breeder said every 1 in 4 phenos will be high cbd.
have you ever grown diesel? do you see the similarities? if its funky desiely fuel with high CBD im all over that shit
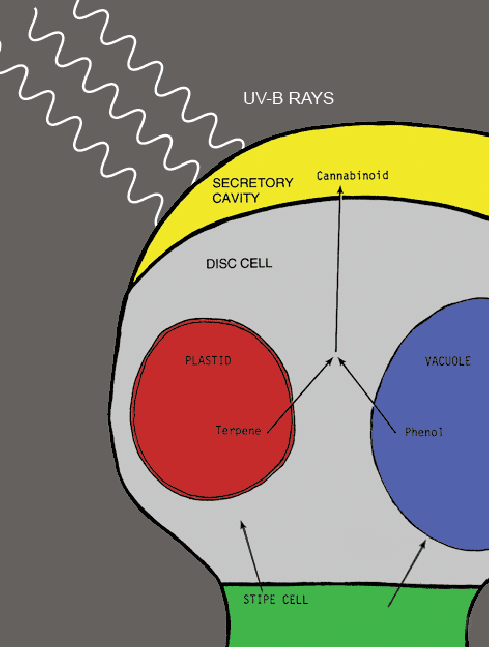
Inside the Trichome
THC and other cannabinoids are produced in only one place on the cannabis plant: inside the heads of the trichomes. How it happens: Organelles produced by the plant called Vacuoles – which contain phenols, a chemical compound similar to alcohol [pictured below in blue], and another type of organelle called plastids – containing hydrocarbons called terpenes [red], make their way up the trichome stalk [green] and combine inside the secretory cavity into a fibrous mat [yellow]. This concentrated mat is hit by UV-B light waves, causing the creation of cannabinoids. Since all of the psychoactive ingredients are produced inside the trichome, these tiny resin hairs have long been sought after by hash and oil makers and can be separated from the plant and harvested in a variety of ways.
______
Cannabinoids
Cannabinoids are a class of chemical compounds which include the phytocannabinoids (oxygen-containing C21 aromatic hydrocarbon compounds found in the cannabis plant), and chemical compounds which mimic the actions of phytocannabinoids or have a similar structure (e.g. endocannabinoids, found in the nervous and immune systems of animals and that activate cannabinoid receptors). The most notable of the cannabinoids is ∆9-tetrahydrocannabinol (THC)—the primary psychoactive compound of cannabis.
Synthetic cannabinoids encompass a variety of distinct chemical classes: the classical cannabinoids structurally related to THC, the nonclassical cannabinoids including the aminoalkylindoles, 1,5-diarylpyrazoles, quinolines and arylsulphonamides, as well as eicosanoids related to the endocannabinoids
Phytocannabinoids
Phytocannabinoids, also called natural cannabinoids, herbal cannabinoids, and classical cannabinoids, are only known to occur naturally in significant quantity in the cannabis plant, and are concentrated in a viscous resin that is produced in glandular structures known as trichomes. In addition to cannabinoids, the resin is rich in terpenes, which are largely responsible for the odour of the cannabis plant.
Phytocannabinoids are nearly insoluble in water but are soluble in lipids, alcohols, and other non-polar organic solvents. However, as phenols, they form more water-soluble phenolate salts under strongly alkaline conditions.
All-natural cannabinoids are derived from their respective 2-carboxylic acids (2-COOH) by decarboxylation (catalyzed by heat, light, or alkaline conditions).
Types
At least 85 cannabinoids have been isolated from the cannabis plant. All classes derive from cannabigerol-type compounds and differ mainly in the way this precursor is cyclized.
Tetrahydrocannabinol (THC), cannabidiol (CBD) and cannabinol (CBN) are the most prevalent natural cannabinoids and have received the most study. Other common cannabinoids are listed below:
(CBG) Cannabigerol
(CBC) Cannabichromene
(CBL) Cannabicyclol
(CBV) Cannabivarin
(THCV) Tetrahydrocannabivarin
(CBDV) Cannabidivarin
(CBCV) Cannabichromevarin
(CBGV) Cannabigerovarin
(CBGM) Cannabigerol Monoethyl Ether
Tetrahydrocannabinol (THC) is the primary psychoactive component of the plant. THC has approximately equal affinity for the CB1 and CB2 receptors.
Delta-9-Tetrahydrocannabinol (Δ9-THC, THC) and delta-8-tetrahydrocannabinol (Δ8-THC), mimic the action of anandamide, a neurotransmitter produced naturally in the body. The THCs produce the high associated with cannabis by binding to the CB1 cannabinoid receptors in the brain.
Cannabidiol (CBD) is not particularly psychoactive in and of itself, and was thought not to affect the psychoactivity of THC. However, recent evidence shows that smokers of cannabis with a higher CBD/THC ratio were less likely to experience schizophrenia-like symptoms. This is supported by psychological tests, in which participants experience less intense psychotic-like effects when intravenous THC was co-administered with CBD (as measured with a PANSS test). Cannabidiol has no affinity for CB1 and CB2 receptors but acts as an indirect antagonist of cannabinoid agonists. Recently it was found to be an antagonist at the putative new cannabinoid receptor, GPR55, a GPCR expressed in the caudate nucleus and putamen. Cannabidiol has also been shown to act as a 5-HT1A receptor agonist, an action which is involved in its antidepressant, anxiolytic, and neuroprotective effects.
It appears to relieve convulsion, inflammation, anxiety, and nausea. CBD has a greater affinity for the CB2 receptor than for the CB1 receptor
CBD shares a precursor with THC and is the main cannabinoid in low-THC Cannabis strains.
Cannabinol (CBN) is the primary product of THC degradation, and there is usually little of it in a fresh plant. CBN content increases as THC degrades in storage, and with exposure to light and air.
Cannabigerol (CBG) is non-psychotomimetic but still affects the overall effects of Cannabis. It acts as an α2-adrenergic receptor agonist, 5-HT1A receptor antagonist, and CB1 receptor antagonist. It also binds to the CB2 receptor.
Tetrahydrocannabivarin (THCV) is prevalent in certain South African and Southeast Asian strains of Cannabis. It is an antagonist of THC at CB1 receptors and attenuates the psychoactive effects of THC.
Cannabichromene (CBC) is non-psychoactive and does not affect the psychoactivity of THC.
Plant profile
Cannabis plants can exhibit wide variation in the quantity and type of cannabinoids they produce. The mixture of cannabinoids produced by a plant is known as the plant's cannabinoid profile. Selective breeding has been used to control the genetics of plants and modify the cannabinoid profile. For example, strains that are used as fiber (commonly called hemp) are bred such that they are low in psychoactive chemicals like THC. Strains used in medicine are often bred for high CBD content, and strains used for recreational purposes are usually bred for high THC content or for a specific chemical balance.
Quantitative analysis of a plant's cannabinoid profile is usually determined by gas chromatography (GC), or more reliably by gas chromatography combined with mass spectrometry (GC/MS). Liquid chromatography (LC) techniques are also possible, although these are often only semi-quantitative or qualitative. There have been systematic attempts to monitor the cannabinoid profile of cannabis over time, but their accuracy is impeded by the illegal status of the plant in many countries.
Plant synthesis
Cannabinoid production starts when an enzyme causes geranyl pyrophosphate and olivetolic acid to combine and form CBG. Next, CBG is independently converted to either CBD or CBC by two separate synthase enzymes. CBD is then enzymatically cyclized to THC. For the propyl homologues (THCV, CBDV and CBNV), there is a similar pathway that is based on CBGV.
History
Cannabinoids were first discovered in the 1940s, when CBD and CBN were identified. The structure of THC was first determined in 1964.
Due to molecular similarity and ease of synthetic conversion, CBD was originally believed to be a natural precursor to THC. However, it is now known that CBD and THC are produced independently in the cannabis plant.
